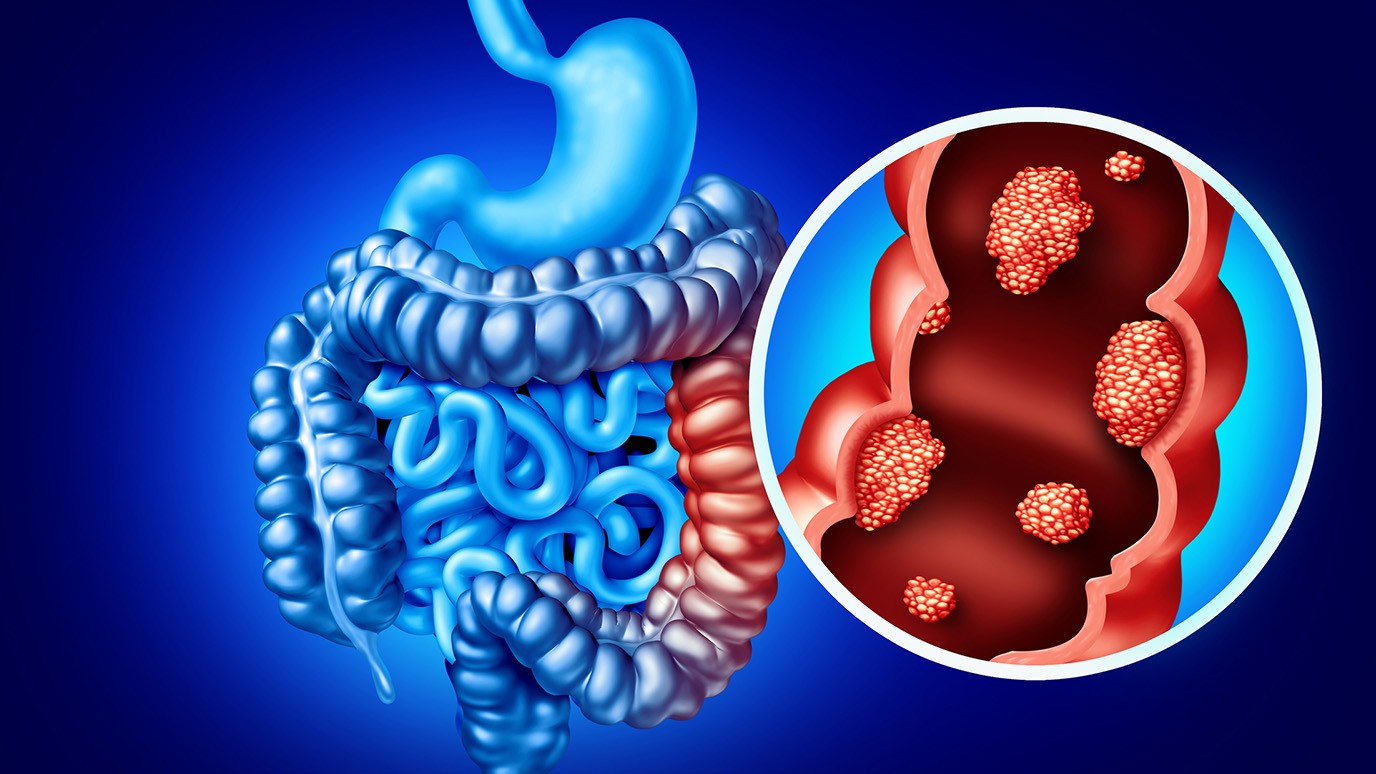
A New Chapter in Prostate Cancer Care: Why England’s Decision on Abiraterone Matters
In January, prostate cancer moved to the centre of the UK health conversation after confirmation that a long-established drug, abiraterone, will now be offered far more widely across England. The announcement sparked extensive coverage, not because the medicine itself is new, but because of what its broader availability represents: a shift in how quickly evidence-based treatments reach patients through the NHS. This trend digest explores how the decision emerged, how it has been framed across the media, and what it could mean for men diagnosed in the coming months.
Main Topic Overview
Abiraterone has been used for years in advanced prostate cancer, particularly for men whose disease has spread. Previously in the 2010s, access was limited to later stages, largely due to cost and commissioning decisions. Building on earlier clinical trials and updated cost assessments, NHS England has now approved its use at an earlier point in treatment for thousands more patients. This is not the first time a cancer drug has been reassessed after prices fell or evidence strengthened, but the scale of the expansion has drawn unusual attention.
News Coverage
Prostate drug, abiraterone, to be offered to thousands in England

The BBC focused on the practical implications of the decision, explaining who will be eligible and how the drug fits alongside standard hormone therapy. The report highlighted estimates that thousands of men each year could benefit, with survival extended by months or years in some cases. Importantly, the BBC placed the announcement within the NHS approval process, emphasising that the decision followed updated guidance and pricing changes rather than a sudden policy shift.
NHS expands access to prostate cancer drug in England to save thousands of lives

The Guardian framed the move as a major public health development, drawing attention to modelling that suggests significant life extension at population level. It also revisited criticism that men in England previously faced a postcode lottery compared with patients elsewhere. By quoting clinicians and patient advocates, the piece underscored how long the change had been anticipated, while remaining careful to describe projected outcomes rather than guaranteed results.
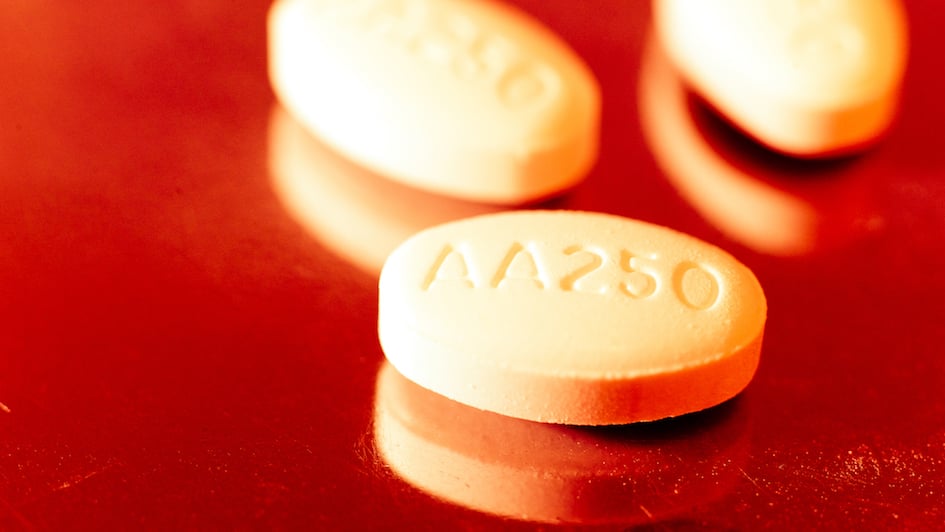
Drug costing just £250 will extend life of prostate cancer patients

The Times emphasised cost, pointing out that abiraterone is now available as a generic medicine. The article argued that the reduced price altered the balance between affordability and benefit, making wider access viable. By focusing on economics, the coverage highlighted a recurring theme in NHS decision-making: treatments sometimes become accessible not because science changes, but because markets do.
Thousands of men to receive prostate cancer drug on NHS

The Telegraph’s reporting concentrated on rollout timelines and eligibility criteria, noting that access could begin within weeks. The article explored how the decision fits into wider NHS cancer targets and early diagnosis efforts. Rather than focusing on emotional narratives, it leaned into service delivery and the scale of implementation across England.
Life-extending prostate cancer drug to be made available to thousands more patients

The Independent highlighted reaction from clinicians and charities, many of whom described the move as overdue. The piece also addressed limitations, noting that abiraterone is not suitable for every patient and must be carefully managed. This balanced tone reflected the outlet’s focus on expectations versus clinical reality.
Thousands more men to be offered life-extending prostate cancer drug on NHS

ITV’s coverage focused on patient impact, using case examples to explain how earlier access could change treatment journeys. It also summarised how the drug works alongside hormone therapy, making the science accessible without oversimplification. The report connected individual experience with national policy.
The ICR welcomes NHS decision to grant lifesaving prostate cancer drug
The Institute of Cancer Research responded from a research perspective, pointing to the trials that underpinned the decision. Its statement traced how long-term evidence accumulation eventually translated into policy. This added historical depth, showing how research timelines and NHS adoption rarely move at the same pace.
Life-extending prostate cancer drug set to be offered to thousands within weeks

The Mirror emphasised speed, stressing that men could see changes quickly rather than over years. The article leaned into accessibility and reassurance, reflecting how the story resonated with families already navigating prostate cancer. While lighter on policy detail, it reinforced the immediacy of the development.
Summary / Insights
Across outlets, a consistent theme emerges: abiraterone itself is not new, but its wider NHS availability is. This isn’t the first time a cancer drug has reached more patients after prices fell and evidence matured, but the coverage shows how strongly access decisions resonate with the public. The likely outcome is a gradual but meaningful shift in standard prostate cancer care in England, shaped as much by economics and policy as by medical discovery.