Wegovy 7.2mg: Why the UK’s Approval of a Stronger Dose Marks a Turning Point in Obesity Care
The UK’s decision to approve a higher-dose version of the weight-loss drug Wegovy has triggered a surge of attention across health policy, clinical practice, and public debate. The newly approved 7.2mg dose—three times stronger than earlier maintenance levels—lands at a moment when demand for injectable weight-loss treatments is already straining supply, reshaping NHS priorities, and redefining how obesity is treated in Britain.
Main Topic Overview
Wegovy, developed by Novo Nordisk, is a once-weekly injection based on semaglutide, a GLP-1 receptor agonist originally used in diabetes care. Lower doses have been available in the UK for weight management, typically topping out at 2.4mg per week. The newly approved 7.2mg dose represents a significant escalation, aimed at patients with severe obesity who have not achieved sufficient results on existing regimens.
This is not the first time obesity drugs have generated excitement in the UK. Previous waves—from appetite suppressants in the 1990s to bariatric surgery expansions in the 2010s—promised breakthroughs but raised questions about access, safety, and long-term outcomes. The current generation of GLP-1 drugs has revived those debates, this time with far stronger clinical evidence and unprecedented public demand.
News Coverage
Weight loss jab to launch stronger dosage as UK demand set to soar

The Independent frames the higher-dose launch as a response to surging UK demand, noting that existing supplies of Wegovy have struggled to keep pace with patient interest. The article highlights how clinicians expect the stronger dose to be reserved for carefully selected cases, rather than broad use. It also situates the approval within a wider shift in how obesity is discussed, increasingly as a chronic medical condition rather than a lifestyle issue. At the same time, it raises questions about whether demand could once again outstrip availability.
Novo Nordisk’s Wegovy higher dose injection approved by UK MHRA for obesity

PMLiVE focuses on the regulatory angle, detailing the MHRA’s decision-making process and the clinical data supporting the 7.2mg dose. The coverage emphasises trial results showing significantly greater average weight loss compared with standard dosing. It also notes that approval does not automatically translate into NHS access, underlining the distinction between regulatory clearance and health-system adoption. This separation is likely to shape the next phase of debate.
Triple-strength fat jab dose gets green light from NHS watchdog as users shed 'significantly more weight'

The Sun adopts a more headline-driven tone, spotlighting dramatic weight-loss outcomes and framing the approval as a breakthrough moment. While the language is more emotive, the underlying reporting echoes other outlets in pointing to stronger results at higher doses. The article also references NHS oversight, reflecting public interest in whether such treatments will be publicly funded. Its framing illustrates how the same regulatory decision can be interpreted very differently across the media landscape.
UK approves new dose of weight loss drug that's three times as powerful
.jpg)
LADbible positions the story for a younger, social-media-savvy audience, focusing on the idea of a dose that is “three times as powerful.” Beyond the framing, it highlights practical questions around who qualifies for the treatment and what side effects may increase with higher dosing. The piece reflects how awareness of medical policy is now spreading well beyond specialist or health-focused outlets. This broader reach may influence public expectations of access.
Stronger dose of weight-loss jab approved for launch in UK
This earlier Independent report provides context on how the approval fits into the UK’s broader obesity strategy. It recalls previous NICE decisions on weight-loss drugs and the cautious expansion of access through specialist services. By placing the 7.2mg dose alongside past policy steps, the article suggests continuity rather than sudden disruption. It reinforces the idea that the approval is part of a longer policy evolution.
MHRA approves Novo Nordisk’s triple-strength Wegovy

Chemist+Druggist zeroes in on implications for pharmacists and prescribing pathways. The article notes how higher dosing may require closer monitoring and clearer guidance at the point of dispensing. It also references earlier supply shortages, reminding readers that regulatory approval does not resolve logistical challenges. For frontline healthcare professionals, the approval raises as many operational questions as clinical ones.
TRIPLE DOSE Fat-Blasting Wegovy Dose Triples to 7.2mg Weekly
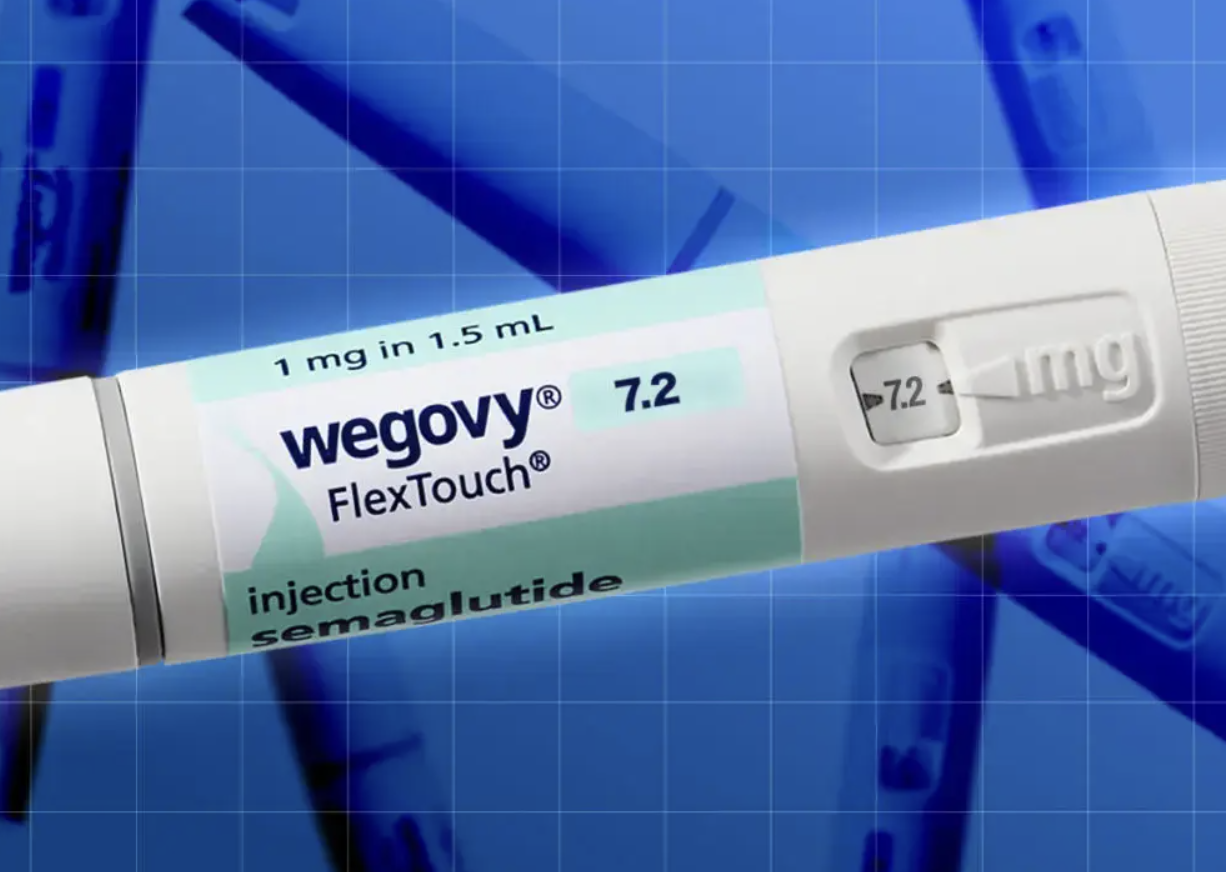
UK News in Pictures presents the approval in highly visual, punchy terms, reinforcing how weight-loss drugs have become a mass-media topic. While lighter on regulatory detail, it underscores the headline fact of a 7.2mg weekly dose and the expectation of stronger results. This style of coverage reflects public fascination with measurable, dramatic outcomes. It also hints at the risk of oversimplifying complex medical decisions.
Summary / Insights
Taken together, the coverage shows broad agreement on the significance of the 7.2mg approval, alongside diverging emphases. Specialist outlets focus on regulation, evidence, and clinical practice, while mainstream and tabloid media highlight dramatic weight-loss potential. Historically, new obesity treatments have often faced a gap between approval and access, and early signs suggest this pattern may repeat. The approval marks a milestone, but it also opens a new chapter of debate about who should receive these drugs, under what conditions, and at what cost.
TL;DR: The UK has approved a much stronger 7.2mg dose of Wegovy, signalling a major shift in obesity treatment. Evidence suggests greater weight loss, but questions remain around access, supply, and NHS funding.